Widespread rough white lesions of the mouth
Can you make the correct diagnosis?
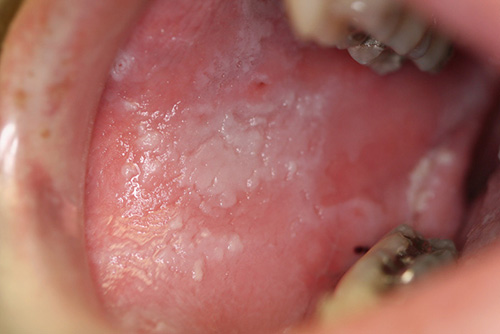
This is a 20-year old white male who presented to his dentist with a 1-year history of widespread white lesions involving several oral sites including the bilateral buccal mucosa, bilateral vestibules and upper and lower lips.
Sorry! you are incorrect
Lichen planus would be a good disease to include on the differential diagnosis given the diffuse nature of this condition and its presence on the bilateral buccal mucosa and vestibule. However, striation and reticular pattern are not present and the patient is on the younger side for LP. The histology is not supportive of LP.
Lichen planus is a common chronic immune mediated disease affecting 2% of the population. It has a variety of clinical presentations, management, and predisposing factors that renders it a family of diseases rather than one entity with a specific treatment modality (1, 2). To be specific, lichen planus can be immune mediated, drug induced, associated with transplant rejection (also known as graft verses host disease), or associated with galvanic effect (when two separate restorations are in opposite contact with each other). It is also seen in families (though rarely), and is known to undergo a malignant transformation in about 2.5% of cases. Given the versatility of this disease, it is difficult to apply one treatment modality for all presentations (2, 3).
The most common type of LP is the chronic T-lymphocyte-mediated disease where CD4 (early stage) and CD8 (later stages) T-lymphocytes are stimulated, releasing lymphokines such as a tumor necrosis factor, which lead to the destruction of the cells in the basal and parabasal cell layers (2). This type of oral LP tends to come and go, sometimes lasting a lifetime. Lichen planus of the skin usually resolves within 1-3 years while only 20% of oral LP cases resolve in that period of time. Lichen planus occurs in adults between 30 and 70 years of age with a strong female predilection (1-3). It is often associated with stressful lifestyles. Skin LP presents as purplish, pruritic papules with a white keratotic surface, commonly on the flexor surfaces of the wrists, trunk and the genitalia. Oral LP presents as keratotic and reticular (the reticular pattern is the most common type), atrophic or erosive (the erosive type is the most important clinically), or plaque-like (the hypertrophic type, which is the least common). Oral LP presents most commonly in a symmetrical manner on the bilateral buccal mucosa, followed by tongue and gingiva. Reticular type LP is asymptomatic and presents with interlacing lines against an erythematosus bluish background; the lines are known as the striae of Wickham. Erosive type LP, on the other hand, is symptomatic—patients complain of sensitivity to hot and cold, spicy, acidic, and alcoholic food and beverages. Erosive LP also presents in locations similar to those of the reticular type. At times, erosive LP can be hard to distinguish from other mucocutaneous diseases such as benign mucous membrane pemphigoid (BMMP) and pemphigus vulgaris (PV). Hyperplastic or plaque LP is uncommon and presents as a confluent white plaque that is often mistaken for leukoplakia. The hyperplastic type is more common on the dorsal surface of the tongue, gingiva, and palate, making a clinical diagnosis of LP difficult at times, especially if the patient smokes. LP can also present on the gingiva alone as a thin and atrophic lesion known as desquamative gingivitis. The clinical differential diagnosis for desquamative gingivitis should include mucous membrane pemphigoid and pemphigus vulgaris.
A discussion of LP would be incomplete without addressing Lichenoid Drug Reaction, a condition that is especially common in elderly patients. It most frequently presents as erosive lichen planus on the bilateral buccal mucosa. It is associated with the ingestion of a number of medications including antibiotics, antihypertensive drugs, allopurinol (gout), diuretics, antidiabetics, gold, mercury, antihistamines and many others. The malignant potential of oral LP is a significant clinical concern, especially in long-term erosive lichen planus patients; transformation in reticular LP has also been documented, albeit rarely so (4). The World Health Organization (WHO) defines oral LP as a precancerous condition (5) with the full understanding that the risk for transformation is only around 2.5%. The locations of transformation are not in the high-risk locations of the conventional oral SCC, but rather involve the buccal mucosa, tongue and gingiva— the same locations as the conventional lichen planus.
Lichen planus is a disease that requires both clinical and histological features to arrive to a definitive diagnosis. Depending on the LP type, the epithelium ranges from thin and erosive to thick and keratotic (2, 3). All types have a band-like infiltrate of T-lymphocytes. The basal cell layer shows evidence of degeneration; rete pegs are or are not present, basement membrane zone is thickened, and cystoid bodies (Civatte bodies) may be present. The immunofluorescence (IMF) features include positive staining with antibody to fibrinogen present along the basement membrane. Treatment ranges from no treatment for the asymptomatic reticular type, to topical steroids, intralesional steroid therapy (rarely used), and systemic steroid therapy (if the condition is severe such as in erosive LP). The best treatment for lichenoid drug reaction is replacement of the causing medication with a substitute. Treatment for transformation would follow the criteria for the treatment of oral squamous cell carcinoma.
Sorry! you are incorrect
Smoker’s keratosis is a reasonable condition to consider in patients with a long history of heavy smoking. It is rare for smoker’s keratosis to be this diffuse. Since this patient smoked 4-5 cigarettes per day for less than three years, it would be unlikely that these lesions are tobacco induced. The histology was also not supportive of smoker’s keratosis.
Smoker’s keratosis should be among the many oral changes associated with the use of tobacco (6), specifically smoked tobacco. In a study in Pakistan which examined the normal gingiva of 16 patients, smokers were found to have more epithelial keratosis in general than non-smokers (7) Smoker’s keratosis is most common in middle-aged males and appears as a white lesion, ranging in texture from smooth to rough or verruciform, usually occurring in the buccal mucosa, mucobuccal fold and/or floor of mouth. Histologically, the lesions range from benign hyperkeratosis and acanthosis to various grades of epithelial dysplasia, and to invasive squamous cell carcinoma. Treatment depends on the histology and the clinical presentation. It may range from cessation of tobacco use (which may lead to the reversal of the white lesions) to surgical or laser excision.
Sorry! you are incorrect
Given the diffuse nature of the white lesions in this patient, Candidiasis should be considered. However, Candidiasis is an opportunistic disease; and this patient did not fit any of the criteria under which Candidiasis occurs in the mouth. Also, the white lesions did not wipe out, which they typically do in psuedomembranous types of Candidiasis. The histology was also not supportive of this disease.
Candidiasis is the most opportunistic infection in the world. If it occurs in non-compromised patients it usually involves the tongue, skin folds, vagina and urinary tract. It may occur in compromised patients especially in the GI tract, lungs, heart valve, kidneys and meninges (8-9). It is caused by Candida albicans, which presents in the form of delicate pseudo hyphae and budding spores. Candida is normally found, in up to 50% of the population, in the oral cavity (8). It affects high-risk patients such as infants under six months of age for lack of immunity, pregnant females, and debilitated patients with chronic diseases, diabetics, patients who are immunosuppressed, and patients on long-term broad-spectrum antibiotics. It is classified into acute and chronic. Acute includes acute pseudomembranous found in chronically ill patients and infants (9). It presents as white, soft, slightly elevated plaques on the buccal mucosa and tongue. It can be wiped out leaving a relatively normal appearing mucosa, or slightly red mucosa. Acute atrophic is erythematous and painful. Chronic includes chronic hyperplastic which is a firm, white, persistent plaque on the lip, tongue and buccal mucosa. It is difficult to wipe this type off. In terms of histology: besides candidal hyphae there is hyperkeratosis, acanthosis and chronic mucositis. Biopsy would show hyphae and spores, and a cytology smear is just as effective. Treatment: Nystatin suspension for oral lesions. If the patient does not respond to treatment, check for diabetes, AIDS, or other endocrinopathies and immune suppressive diseases (8-9).
Congratulations! You are correct
Cannon first coined the term in 1935 (11). Today, it is identified as a genetic disease caused by a point mutation in cytokeratin types 4 and 13 (12). This is a rare hereditary autosomal dominant condition that predominantly affects the non-keratinized mucosa including the oral cavity, nose, esophagus, vagina and rectum. The oral mucosa is more frequently affected. It is of variable penetrance (10). Although it typically occurs before age 20, it may occur at birth or during childhood or late adolescence, as is the case with this patient (12). White sponge nevus can be mistaken for other types of leukoplakias. The history of the presence of these lesions since childhood combined with a family history of the disease and the histology are adequate in rendering a definitive diagnosis (12). This patient’s mother and one of her cousins have similar lesions. Clinically, it presents as white, folded plaques that can peel away. The spongy mucosa can look translucent simulating leukoedema. It can occur anywhere in the oral cavity but most commonly on the buccal mucosa followed by the lips, floor of mouth and the alveolar ridge (10). Histologically, the epithelium is thick and is covered by a thick and shaggy parakeratin with keratin plugging and intracellular edema of the spinous layer. Condensations around the nuclei are present in the upper spinous layer (Fig 4). No treatment is necessary. It has an excellent prognosis.
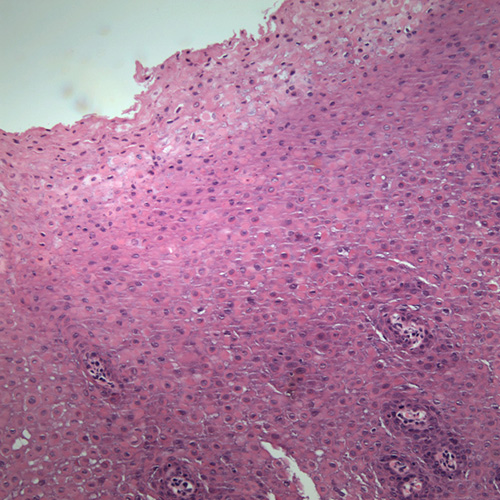
Figure 4. Low power (x100) histology shows a fragment of oral mucosa with thick spinous layer covered by a thick layer of parakeratin. Note the epithelial cells throughout the spinous layer especially in the superficial layers exhibiting keratin condensations around the nuclei.
Treatment
The buccal mucosa was biopsied under local anesthesia. An elongated piece of about 8 X 4 X 4 mm in greatest dimensions was removed and submitted for microscopic examination. The area healed within a short period of time. Based on the results of the incisional biopsy, the patient was advised against removal of the remaining lesions. No further treatment was rendered.
References
- Silverman S, Gorsky M et al. A retrospective study of findings and management in 214 patients with oral lichen planus. Oral Surg Oral Med Oral Pathol 1991; 72:665–670.
- Chiappelli F, Kung MA et al. Cellular immune correlates of clinical severity in oral lichen planus: preliminary association with mood states. Oral Dis. 1997; 3:64-70.
- Silverman S. Oral lichen planus: a potentially premalignant lesion. J Oral Maxillofac Surg 2000; 58:1286–1288.
- Duffey DC, Eversole LR et al. Oral lichen planus and its association with squamous cell carcinoma: an update on pathogenesis and treatment implications. Laryngoscope. 1996; 106:357-362.
- Pindborg JJ, Reichart PA et al. WHO International histological Classification of Tumours. Histological typing of cancer and precancer of the oral mucosa. , Springer, Berlin (1997).
- Taybos G. Oral changes associated with tobacco use. Am J Med Sci. 2003 Oct;326(4):179-82.
- Rahman BU, Rahman MM, Arslan A. The effects of cigarette smoking on human gingival tissues (a histopathological study). J Pak Med Assoc. 1994 Sep; 44(9):210-2.
- Sherman RG, Oral candidiasis. Quintessence Int. 2002 Jul-Aug; 33(7):521-32.
- Lynch DP.Oral candidiasis. Oral Surg Oral Med Oral Pathol. 1994 Aug;78(2):189-93.
- Martelli H Jr Pereira SM, et al. White sponge nevus: report of a three-generation family. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007 Jan; 103(1):43-7.
- A.B. Cannon, White sponge nevus of the mucosa (nevus spongious albus mucosa), Arch Dermatol Syphilol 31 (1935), pp. 365–370.
- Shibuya, J. Zhang, S. Yokoo, M. Umeda and T. Komori, Constitutional mutation of keratin 13 gene in familial white sponge nevus, Oral Surg Oral Med Oral Pathol Oral Radiol Endod 96 (2003), pp. 561–565