July 2010: Blue Hard Palate
Can you make the correct diagnosis?
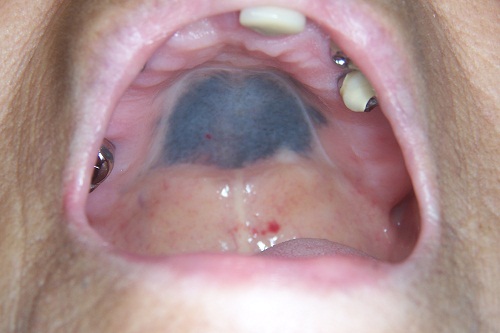
This is a 78-year-old white female who has had a uniformly blue lesion in her hard palate for over two years. It is otherwise asymptomatic and is not present anywhere else.
Sorry! you are incorrect
Dark blue to black and flat lesions of the mouth are usually caused by endogenous (melanin, blood, hemosiderin) or exogenous (metal, carbon) particles. Melanocytic nevi are usually brown to black and sometimes blue as is the case with blue nevus. Oral pigmented nevi, including blue nevi, tend to be small—around 6 mm or smaller in size. This lesion is very large and generalized, therefore unlikely to be a nevus despite being in the right location and having the uniform blue color. Neither the size nor the histology in this case are supportive of a diagnosis of nevus.
Melanocytic nevi (common moles) are rare in the mouth but are very common on the skin. Most skin nevi are of the acquired (developmental) type while congenital nevi are much less frequent on the skin and also far less frequent in the mouth. Nevi are benign and most likely hamartomatous proliferations of cells of neural crest origin which can be melanocytes or other closely related cells. In the oral cavity, they most commonly occur on the hard palate, buccal mucosa, gingiva and lips (1-5). They are more common in females around 30 years of age. Intramucosal (intradermal) and blue nevi constitute 80-90% of all oral melanocytic nevi with compound and junctional nevi accounting for less than 10-20% of cases (1-5). Nevi vary in color from colorless to dark blue, brown or black; some oral nevi lack the brown color. They can be flat, raised, smooth-surfaced or papillary with a smooth border and symmetrical morphology. Histologically, they vary in morphology depending on the type. Nevi are usually not removed unless they are chronically irritated. They are sometimes removed for aesthetic reasons. They should be removed if they change in appearance in accordance with the “A-E rule”: A) for asymmetry, B) for border (irregular border), C) for color (lack of uniformity in color), D) for diameter (larger than six mm in diameter) and E) for enlarging (5). They should be surgically removed if they ulcerate or bleed.
Congratulations! You are correct
Two pigments were identified in the incisional biopsy from this patient: melanin and hemosiderin. Melanin was identified in the connective tissue without nevus cells or melanocytes in the area and hemosiderin was also identified in the connective tissue without any vascular lesion or known cause of hemorrhage.
Many drugs are known to cause skin and mucosal hyperpigmentation as a secondary effect (6-8). The mechanism by which pigmentation is achieved varies from direct stimulation of the melanocyte activity to deposition of pigmented drug metabolites. Drugs that are known to cause mucosal and skin pigmentation including minocycline (8), tetracycline, antimalarial agents (particularly chloroquine and hydroxychloroquine), chlorpromazine, birth control pills, AZT, and ketoconazole. The pigmented lesions that are secondary to drug use can be generalized or localized or can be small or large in size. The lesions are flat and range in color from tan to dark brown. Our patient was on Plaquenil (hydroxychloroquine), which falls under the category of antimalarial drugs (6-7) but is also used to treat diseases such as rheumatoid arthritis. A well-known side effect of Plaquenil is skin and mucosal discoloration as well as eye pigmentation. In the mouth, the hard palate seems to be a more common location for discoloration with antimalarial drugs (6-7).
Birth control pills, like pregnancy, can cause discoloration (melasma, mask of pregnancy) of the skin (commonly) and the oral mucosa (less commonly). The use of birth control pills and hormone replacement therapy can have the same effect as pregnancy; this is especially true in olive-skinned females. Pregnancy and estrogen intake leads to melanocyte activity, releasing more melanin especially when the skin is exposed to sun. The brown pigment is mostly on the middle of the face but can involve the skin on the chin and the forehead. The macules can range from a few mms to 2 cm in size. In the mouth, the discoloration is usually linear and present on the anterior gingiva, especially the lower anterior gingiva (similar to smoker’s melanosis). The discoloration disappears months after delivery or the discontinuation of the hormonal use. The discoloration is enhanced when the patient is also a smoker.
Tetracycline, especially its semisynthetic derivative minocycline, has been implicated in oral soft and hard tissue discoloration since 1984. Minocyline is used for the treatment of inflammatory acne and is known for its broad spectrum activity against aerobic and anaerobic gram-negative and gram-positive organisms. Oral mucosal and hard tissue pigmentation is described in up to 4% of long-term users (over eight months), especially at doses of 100 or more milligrams per day (8). Hyperpigmentation associated with chronic steroid use has also been documented.
The second pigment that contributed to the blue color in this case was hemosiderin, which was interpreted to be the result of an old hemorrhage. There is no clinical reason for the hemorrhage, i.e. trauma or negative pressure. We interpreted the hemorrhage to also be drug induced since the patient is on Plavix, which is known to cause easy bruising and bleeding.
References
- Meleti M, Vescovi P, Mooi WJ, van der Waal I. Pigmented lesions of the oral mucosa and perioral tissues: a flow-chart for the diagnosis and some recommendations for the management. Oral Surg Oral Med Oral Pah Oral Radiol. 2008 May;105:606-16.
- Buchner, P.W. Merrel and W.M. Carpenter, Relative frequency of solitary melanocytic lesions of the oral mucosa, J Oral Pathol Med 33 (2004), pp. 550–557.
- Eisen, Disorders of pigmentation in the oral cavity, Clin Dermatol, 2000,18: 579–587.
- Çiçek and Ü. Ertas¨, The normal and pathological pigmentation of oral mucous membrane: a review, J Contemp Dent Pract 2003, 3: 76–86.
- Healsmith, MF, Bourke, JF et al. An evaluation of the revised seven-point check list for the early diagnosis of cutaneous malignant melanoma. Br J Dermatol 1994; 130: 48-50.
- Lerman MA, Karimbux N, Guze KA, Woo SB. Pigmentation of the hard palate. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:8-12.
- Kleinegger CL, Hammond HL, Finkelstein MW. Oral mucosal hyperpigmentation secondary to antimalarial drug therapy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90:189-194.
- Treister NS, Magalnick D, Woo SB. Oral mucosal pigmentation secondary to minocycline therapy: report of two cases and a review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97:718-725.
- Buchner A. Amalgam tattoo (amalgam pigmentation) of the oral mucosa: clinical manifestations, diagnosis and treatment. Refuat Hapeh Vehashinayim. 2004 Jul;21(3):25-8.
- Owens BM, Johnson WW, Schuman NJ. Oral amalgam pigmentations (tattoos): a retrospective study. Quintessence Int. 1992 Dec;23(12):805-10.
- Shah, G, Alster, TS. Treatment of an amalgam tattoo with a Q-switched alexandrite (755 nm) laser. Dermatol Surg 2002; 28: 1180-1181.
- Hicks MJ, Flaitz CM. Oral mucosal melanoma: epidemiology and pathobiology. Oral Oncol. 2000 Mar; 36(2):152-169.
- Rapidis, AD, Apostolidis, C et al. Primary malignant melanoma of the oral mucosa. J Oral Maxillofac Surg. 2003; 61: 1132-1139.
- Rigel, DS, Friedman, RJ et al. The incidence of malignant Melanoma in the United States: issue as we approach the 21st century. Am J Acad Dermatol 1996; 34: 839-847.
Sorry! you are incorrect
A flat and dark blue stain should remind the clinician of an amalgam tattoo; therefore, it was included on the differential diagnosis. However, the size in this case is unusual for an amalgam tattoo unless the tattoo is deliberately created (of course, this patient’s was not), and the histology did not indicate an amalgam tattoo.
Amalgam tattoo is second only to racial pigmentation in being the most common pigmented lesion in the oral cavity. It is an example of a local deposition of exogenous metallic material from a dental filling, usually the silver metal found in an amalgam (9-11). The fine dark granules of this material are deposited on the very thin collagen fibers of the connective tissue, around small blood vessels, of the basement membrane, between muscle fibers, salivary gland tissue and fat. Amalgam tattoo occurs as a result of metal implantation in lacerated mucosa during a dental filling or the removal of an old amalgam filling (9-11). It can also be the result of amalgam falling in the dental socket following an extraction or in the area of an apicoectomy sealing. Like any cutaneous tattoo, it becomes part of the live tissue and leads to a permanent coloration of the mucosa, usually flat and grey to bluish-black or black in color. Occasionally, it can be exophytic or nodular (9); this is usually the result of a foreign body reaction to the metal. The margins can be irregular, well defined or diffuse, blending in with the surrounding unaffected mucosa. The borders are usually flat and not rolled. They can enlarge and give the impression of lateral spread; this happens during the first months of implantation, especially when a foreign body reaction with macrophages presents, carrying the material laterally and giving the impression of spreading, thus making one think beyond amalgam tattoo to a more sinister disease. Once the area scars, the lateral spread ceases. Amalgam tattoo most commonly occurs on the alveolar mucosa, buccal mucosa, and gingiva (9-10), but has been described on the palate, floor of mouth and tongue. It can be viewed radiographically, especially within a socket if present in clusters. Histologically, it presents as granular black material deposited on the surrounding thin collagen fibrils. Sometimes, foreign body reaction with foreign-body-type giant cells, chronic inflammation and scarring are present. Sometimes, an incisional biopsy is recommended to eliminate the possibility of other, more serious diseases; otherwise, there is no need for treatment.
Sorry! you are incorrect
A flat, diffuse dark lesion on the palate is a common presentation for a malignant melanoma. However, the uniformity in the discoloration of this lesion is not supportive of MM; neither is the blue color. Malignant melanomas usually present with a lack of color uniformity, and the color is usually in the family of brown. Uniform blue discoloration is highly unusual for MM. The histology is also not supportive of malignant melanoma.
Malignant melanoma (MM), whether on the skin or in the oral cavity, is of melanocytic origin. Despite the common cell origin, these lesions have different clinical behavior. Mucosal melanomas are very rare and are more aggressive than those of the skin. They usually affect adults with an average age of 55; occasionally, they affect patients under 30 years of age (12-13). Skin MM used to have a poor prognosis but that has changed significantly; in 1950, the 5-year survival rate for skin MM was 50%, while today it is 90% (12-14). However, mucosal melanomas still carry a dismal 5-year survival rate of not higher than 38% (12). Skin MM, like mucosal melanoma, occurs mostly in adults around 50 years of age and rarely affects children.
Cutaneous malignant melanoma is the third most common skin epithelial malignancy (following basal cell carcinoma and squamous cell carcinoma) but by far the most aggressive of the three. In the United States alone, over 50,000 cases are diagnosed annually with about 8,000 dying of the disease each year (12). It has been on the increase since 1960 and has recently plateaued. Australia has the highest number of skin MMs, a number that has been doubling every 10 years (14). The etiology of MM and its rate of increase are complex and are beyond the scope of this discussion. However, it is important to mention that multiple factors seem to play a role the development of the MM and its progression. The most important factor is exposure to ultraviolet light, which explains cases on the skin but does not explain the oral, nasal, and other non-sun exposed areas that develop MM. It has been suggested that the age of the individual at the time of exposure is important (12-14). It is also suggested that sunburns, especially blistering sunburns in childhood, increase the risk of developing MM in adulthood. Genetics play a significant role in MM, especially in patients born in families with a history of this disease. For instance, patients from families with a history of dysplastic mole syndrome or patients who have a first relative with malignant melanoma are at a higher risk of developing the disease (12-14). 70% of cutaneous MMs occur in a preexisting pigmented nevus. Pigmented nevi are common and have a one-in-a-million chance of transformation to MM. This does not necessitate surgical removal, but rather suggests that practitioners should be attentive to alterations in nevi such as changes in color, shape and size; ulceration and/or bleeding; inflammation; pain; and a diameter change of greater than 7mm. These are the clinical criteria used in England (5) to assess the clinical presentation of MM. Bleeding and ulceration of a pigmented nevus should be viewed seriously, and the lesion should be soon biopsied or completely removed and submitted for microscopic evaluation. The clinical features of MM in the United States follow the ABCDE rule: asymmetry, borders, color, diameter and enlargement. A pigmented lesion that is asymmetrical with irregular borders (especially borders with notching) and uneven coloration, is larger than 6 mm in diameter, and continues to enlarge should be viewed with suspicion and biopsied immediately.
The oral mucosa is rarely affected by MM and for that reason, the etiology of oral malignant melanoma is still unclear. Local melanosis and tobacco use have been implicated. Race also appears to play a role; for example, 7.5% of all reported oral melanomas affect those of Japanese descent and 10% affect those of Ugandan descent, compared with less than 1% of cases occurring in Caucasians. Cutaneous MM, however, occurs at a much higher rate in Caucasians than in those with brown or black skin, with a rate of 1:83 in whites compared to 1:1176 in dark-skinned individuals (13). Mucosal melanomas are extremely rare compared to cutaneous melanomas; they constitute less than 1% of all MMs. For that reason, it is always prudent to first rule out metastatic MM to the oral cavity from the skin when a biopsy of an oral pigmented lesion confirms the diagnosis of MM. 80% of oral MMs affect the palate and the maxillary gingiva. The mandibular gingiva can be affected, but rarely. Cutaneous MMs occur mostly on the interscapular area of the back and back of the legs (12). 25% occur in the head and neck area, including the face, eyes, nose and mouth (14). In the oral cavity, they present as flat, multiple, graded or with uneven coloration, and irregular borders; they are sometimes ulcerated. They are usually asymptomatic; pain is described only rarely.
Histologically, MMs demonstrate radial and vertical growth patterns. The vertical pattern is important because it plays an important role in predicting prognosis; for that reason, the depth of invasion is always reported. Depth of invasion in skin MMs is fully established following Clark and Breslow’s staging according to depth (12). Obviously, the deeper the tumor invasion, the higher the microscopic staging and the worse the clinical behavior. Determining prognosis is not simple, though; other factors play a role in worsening the prognosis, such as the location, age and gender of the patient. Surgery is the treatment of choice treatment for MM of the oral cavity. Radiation and chemotherapy may be used in advanced cases.