Non-healing ulcer, lateral border of tongue
Dolphine Oda, BDS, MSc
doda@u.washington.edu
Can you make the correct diagnosis?
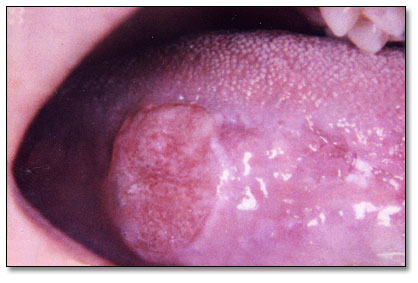
This is a 20-year-old female who was in good health until approximately April 2005 when she noticed a sore on her right lateral tongue.
Sorry! you are incorrect
The oral cavity is frequently afflicted with surface ulcerations: single and multiple, superficial and deep, acute and chronic, of local and of systemic origin. For that reason, it is very important that clinicians view oral ulceration with some attention to etiology. It is important to state that most single oral ulcers are trauma induced and can occur at any age. The source of trauma can be a sharp tooth, an ill-fitting denture or a sharp denture edge, a broken filling, a bite, or any other similar irritant. Chronic single ulcers, especially those associated with dentures, may have rolled borders because of scarring occurring during the healing process. The rolled border-type ulcers are frequently mistaken for SCC. Elimination of the source of trauma will help healing of the ulcer within two to three weeks. Sometimes, however, the source of trauma is difficult to identify, and even when identified and removed, the ulcers do not heal and may remain persistent for months. A biopsy of a non-healing ulcer is warranted for two reasons: to rule out malignancy, such as squamous cell carcinoma, and to initiate healing. This is especially true with ulcers on the lateral border of the tongue, which happens to be a common location for traumatic ulcers. It is good practice to refer patients with ulcers of the mouth when they fail to heal within 2-3 weeks (16). The location in is this case (Fig 1) is consistent with traumatic ulcer, but the histology is not supportive of that diagnosis.
Sorry! you are incorrect
These ulcers are also known by other names, including eosinophilic ulcer, eosinophilic granuloma of the tongue (which is not related to eosinophilic granuloma of the bone as in histiocytosis X disease), and traumatic ulcerative granuloma with stromal eosinophilia (TUGSE). Traumatic granuloma commonly occurs on the dorsal and lateral tongue in patients with a history of trauma. It may affect patients of all ages, including infants (Riga-Fede disease occurs during the first year of life as a result of chronic trauma in the sublingual area to the baby’s lower incisors during breastfeeding). Traumatic granuloma tends to occur more in males than females. Histologically, these ulcers are deep lesions involving the underlying muscle, which may explain the process of slow healing and the tissue eosinophilia. Healing may take up to eight weeks. Eosinophils are found in areas of muscle damage. Clinically, traumatic granulomas tend to be deep ulcers with rolled borders mistaken for SCC (17). Like traumatic ulcer, the location is consistent with traumatic granuloma while the histology is not supportive of it.
Congratulations! You are correct
The focus of this discussion is on oral SCC in individuals under the age of 40, referred to as oral SCC in the “young population.” The “older population” consists of those over 40.
Oral SCC is a highly aggressive neoplasm that currently ranks as the fifth most common malignant neoplasm worldwide and accounts for an estimated 90% of all oral malignancies (1-3). It predominantly affects males over the age of 40 years, with an observed male to female ratio of 2:1 generally and 1.4:1 in the United States (1-2). The most common sites of occurrence are the tongue, the ventral and lateral surfaces, and the floor of mouth. The tongue is also the most common site for oral SCC in the young population. Like most malignancies, the prognosis of oral SCC is dependent on the clinical stage of the disease (1-10).
The incidence of oral squamous cell carcinoma in patients under the age of 40 constitutes 0.4-8.3% of all oral SCC cases and is on the increase (2-3, 10). The prognosis of oral SCC in young patients as compared to the typical older male patient has been the subject of academic discussion for many years with little or no consensus as to its clinical behavior and prognosis. It is generally believed that oral SCC in young patients has a more aggressive behavior and therefore worse prognosis than that in the older population (2-5). For example, Sakaria et al (4) demonstrated a higher recurrence rate of oral SCC in young patients compared to that in older patients. In addition, Hannah et al (5) demonstrated that young and non-smoking females with oral SCC have a 65% recurrence rate, compared to a 41% recurrence rate in older patients. They also demonstrated that oral SCC in young females has a tendency for early recurrence—14 months after removal, as opposed to 40 months in the older population.
However, there are reports that claim that oral SCC in young patients has a better prognosis (6-7). For example, Yamazaki et al (6) reported that patients older than 65 years of age have a worse prognosis than the younger population. This observation was supported by Siegelmann-Danielli et al (7).
Yet a third group of investigators (this author included) assert that the prognosis and survival rate of oral SCC in young and old groups are similar when compared stage-to-stage (2, 13-14). A recently published study by Sasaki et al (2) reported on 35 patients under the age of 40 with oral SCC. The young patient group consisted of 20 males and 15 females; one patient was under 20 years of age, thirteen were ages 21-30 and twenty-one were ages 31-40. The control older patient group comprised 46 females and 64 males: sixteen patients were ages 41-50, twenty-nine were ages 51-60 and sixty-five were over 60. Their findings included similarities in a male-to-female ratio being 1.3:1 in both groups. They also found similarities in most common location which is tongue in both age groups. The size of the primary SCC was statistically significant (P = 0.027), with the older group having a greater number of patients with large (T4) lesions. However, there were no significant differences in the regional lymph node involvement and the overall TNM staging. There was a statistical difference (P < 0.001) in the histologic differentiation of the SCC: most of the SCCs in young patients were well differentiated (65.7%) compared to 32.7% of the SCCs in the older population.
Understanding the mechanism of oral SCC in the young population is important; some suggest that it is a distinct disease entity, on the basis of different biological behavior and etiological factors (2–5, 8-13). Tobacco use, both in cigarette and smokeless forms, is widely known to be strongly associated with oral SCC of the older population; it is a factor in up to 90% of cases (1-13). Some studies report that smoking is not a significant risk factor in the younger population with oral SCC (3, 8, 11-12). However, Llewellyn et al (9) demonstrated that smoking before age 16 and excessive drinking are significant risk factors in the oral SCC in young population. Other proposed risk factors include human papilloma virus, especially type 16 to be more prevalent in the tonsillar and laryngeal carcinomas (11) and other parts of the oral cavity (12). The variety of opinions is a testament to the fact that more epidemiological studies are needed. Long-term prospective clinical studies in large populations and genetic studies are necessary to arrive at a more reliable conclusion.
Determination of the prognosis of oral SCC in both age groups is based on the clinical stage and histologic classification. Although oral SCC is a diagnosis made by histology, surgeons tend to depend exclusively on the TNM classification system for the clinical staging and treatment (1, 11-12). Oral SCC is graded according to the degree of the neoplastic epithelial cell differentiation as described by Broders (15), suggesting that the more differentiated the histology, the better the prognosis. This is not actually the case clinically, since prognosis is more dependent on the TNM staging system. Histological grading ranges from well to moderately to poorly differentiated. The histology of this case was moderate differentiation.
In general, the more posterior the oral SCC, the worse the prognosis (1, 3). The most important prognostic sign is the presence or absence of metastases at the time of diagnosis. The prognosis thus improves when the lesion is detected early. Oral SCC patients die mainly of infection due to lowered resistance or of hemorrhage if the tumor erodes through one of the main blood vessels.
Treatment
Under general anesthesia, the patient underwent a right partial glossectomy (Fig 2) and selective neck dissection involving levels 1, 2, and 3 lymph nodes. During the surgery, a 1.5 cm lesion was noted on the right lateral border of tongue. The patient healed well without complications. She suffered fatigue and lost 15 pounds; she recently regained 5 pounds. She has noted some neck stiffness, but has minimal pain in the tongue. She had some tongue swelling, which caused her speech to be slurred, but that has improved slowly. She denies dysphagia or odynophagia. Postoperative post-ganglionic Horner’s syndrome was also diagnosed, which was causing right side myosis and ptosis of the upper lid, and upside-down ptosis of the lower lid. It was deemed to be most likely secondary to the neck lymph node dissection for squamous cell carcinoma. Surgery has been scheduled to repair the ptosis in her right eye. A four month follow-up visit revealed a fully healed tongue with some scarring and a partial defect but otherwise functional tongue. (Fig 3)
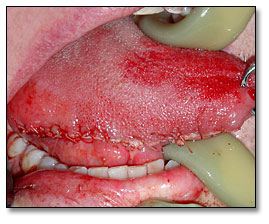
Figure 2. This is a post-surgical photograph demonstrating a sutured and swollen tongue after hemiglossectomy procedure.
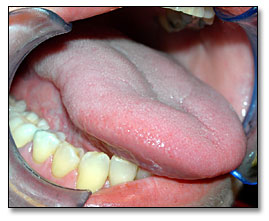
Figure 3. This is a four post- surgical photograph demonstrating a fully healed tongue with some defect and scarring, but otherwise functional.
References
- Bundgaard T, Bentzen SM, Wildt J, Sorensen FB, Sogaard H, Nielsen JE. Histopathologic, stereologic, Epidemiologic, and clinical parameters in the prognostic evaluation of squamous cell carcinoma of the oral cavity. Head & Neck. 1996; 18:142-152.
- Sasaki, T., D. R. Moles, et al. (2005). “Clinico-pathological features of squamous cell carcinoma of the oral cavity in patients <40 years of age." J Oral Pathol Med 34(3): 129-33.
- Durazzo MD , de Araujo CE, Brandao Neto Jde S, Potenza Ade S, Costa P, Takeda F, Bianchi C, Tavares MR, de Britto e Silva Filho G, Ferraz AR. Clinical and epidemiological features of oral cancer in a medical school teaching hospital from 1994 to 2002: increasing incidence in women, predominance of advanced local disease, and low incidence of neck metastases. Clinics. 2005;60:293-298.
- Sarkaria JN, Harari PM. Oral tongue cancer in young adults less than 40 years of age: rationale for aggressive therapy. Head Neck 1994; 16: 10711.
- Hannah V, Karen TP, Jonas TJ, Lisa TG. More aggressive behavior of squamous cell carcinoma of the anterior tongue in young women. Laryngoscope 2000; 110: 16236-6
- Yamazaki H, Inoue T, Koizumi M, Yoshida K, Kagawa K, Shiomi H, Imai A, Tanaka E, Teshima T, Nakamura H, Shimizutani K, Furukawa S, Fuchihata H, Inoue T.. Age as a prognostic factor for late local recurrence of early tongue cancer treated with brachytherapy. Anticancer Res. 1997;17(6D):4709-12
- Siegelmann-Danieli N, Hanlon A, Ridge JA, Padmore R, Fein DA, Langer CJ. Oral tongue cancer in patients less than 45 years old: institutional experience and comparison with older patients. J Clin Oncol 1998; 16: 745-53.
- Franceschi S, Barra S, La Vecchia C, Bidori E, Negri E, Talamini R. Risk factors for cancer of the tongue and the mouth. A case-control study from northern Italy. Cancer 1992; 70: 2227-33.
- Llewellyn CD, Johnson NW, Warnakulasuriya KA. Risk factors for squamous cell carcinoma of the oral cavity in young people a comprehensive literature review. Oral Oncol 2001; 37: 401-18.
- Schantz SP, YU GP. Head and Neck cancer incidence trends in young Americans, 1973-1997 with a special analysis for tongue cancer. Arch Otolaryngol Head Neck Surg 2002; 128: 268-327.
- El-Mofty SK, Lu DW. Prevalence of human papillomavirus type 16 DNA in squamous cell carcinoma of the palatine tonsil, and not the oral cavity, in young patients: a distinct clinicopathologic and molecular disease entity. Am J Surg Pathol. 2003; 27(11):1463-1470.
- Scully C. Oral cancer; the evidence for sexual transmission. Br Dent J. 2005; 199:203-207.
- Manuel S, Raghavan SK, Pandey M, Sebastian P. Survival in patients under 45 years with squamous cell carcinoma of the oral tongue. Int J Oral Maxillofac Surg. 2003; 32:167-173
- Mathew Iype E, Pandey M, Mathew A, Thomas G, Sebastian P, Krishnan Nair M. Squamous cell carcinoma of the tongue among young Indian adults. Neoplasia. 2001; 3:273-277.
- Broders AC. Squamous-cell epithelioma of the lip: a study of five hundred and thirty-seven cases. JAMA. 74(10):656-64 (1920).
- Houston, G. traumatic ulcers. E-Medicine.com, updated July 02
- Alawi, F. Eosinophilic Ulcer. E-Medicine.com. Updated May 02
- Shah A, E-Medicine.com. Updated July 03
Sorry! you are incorrect
The oral cavity is a site where a number of acute and chronic infectious diseases occur as a local disease or as a manifestation of a systemic disease. They include diseases of viral, fungal and bacterial origin. They usually present in multiple or in a diffuse manner ranging from ulcers to small and nodular lesions to verrucoid lesions. Rarely would infectious diseases present as a single ulcer, simulating an OSCC. A primary syphilitic chancre can present like that. Others would include ulceration induced by mycobacterium tuberculosis, deep fungal infection such as blastomycosis and histoplasmosis. Oral manifestations of tuberculosis and histoplasmosis are unusual without lung involvement and a disseminated disease and occur more in immune compromised patients. Gingiva and the mandibular vestibule are more common locations but tongue can occasionally be affected. Chancre can occur in any location, including the tongue, lips and hard palate. As mentioned previously, single oral ulcers of infectious disease origin are a rare occurrence and should be kept low on the differential diagnosis list (18). The histology in this case was not supportive of an infectious process.