Multilocular Expansile Radiolucency, Posterior Mandible
Can you make the correct diagnosis?
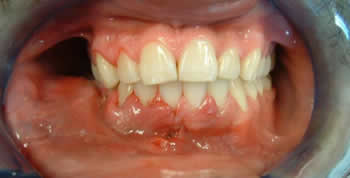
This is a 26-year-old male who is a recent immigrant from Mexico. He presented for the evaluation of a right mandibular swelling of two years’ duration which has become progressively larger during that time.
Sorry! you are incorrect
Odontogenic keratocyst is an aggressive cyst known for its rapid growth and its tendency to invade the adjacent tissues, including bone. It has a high recurrence rate and is associated with bifid rib basal cell nevus syndrome (1, 2). The majority of patients are in the age ranges of 20-29 and 40-59, but cases in patients ranging in age from 5 to 80 years have been reported. The distribution between sexes varies from equal distribution to a male-to-female ratio of 1.6:1, except in children (1, 2). Odontogenic keratocyst predominantly affects Caucasian populations and, if one may judge from the limited evidence provided by the literature, is chiefly of Northern European descent (1).
Odontogenic keratocysts may occur in any part of the upper and lower jaw, with the majority (almost 70%) occurring in the mandible. They occur most commonly in the angle of the mandible and ramus (1). Posterior mandible is an area common to many benign odontogenic tumors such as ameloblastoma and is also a typical location for dentigerous cysts. Radiographically, OKCs present predominantly as unilocular radiolucencies with well-defined, sclerotic or scalloped borders. They may also present as multilocular radiolucencies. Odontogenic keratocysts of the maxilla are smaller in size when compared to those occurring in the mandible; larger OKCs tend to expand bone, but mildly—obvious clinical expansion (which is the case in this patient) should be viewed with suspicion for a neoplasm. OKCs can also present as small and oval radiolucencies between teeth simulating a lateral periodontal cyst, in an area of an extracted tooth simulating a residual cyst, at the apex of a vital tooth mistaken for a periapical cyst, or in the anterior maxilla between the central incisors simulating an incisive canal cyst (2). OKCs grow to sizes larger than any other odontogenic cysts. They usually penetrate the bone rather than expand it and grow in an anterior to posterior direction (3). Despite this aggressive growth, they often remain asymptomatic, thus growing to large sizes and hollowing the bone.
Odontogenic keratocysts are significant clinical entities due to their tendency for recurrence and destructive behavior. They are known to have a high recurrence rate, ranging from 13% to 60% (1, 2). Complete surgical removal is the treatment of choice. Surgery includes enucleation, curettage, enucleation and peripheral ostectomy, and resection depending on the radiographic presentation, location and clinical behavior. Surgery combined with Carnoy’s solution or liquid nitrogen treatment has been effective in reducing recurrence rate (1, 2). At times, adjacent or associated teeth are extracted in the interest of complete removal. Some investigators advocate marsupialization and occasionally resection of the more aggressive cysts that tend to perforate buccal and lingual bone. Resection is a rare modality of treatment. Most cysts recur within the first three years while others may recur as late as after 16 years (1, 2). Conservative surgical removal and long-term follow-up is the treatment of choice by most clinicians. The histology of this case is not supportive of odontogenic keratocyst.
Sorry! you are incorrect
Ameloblastoma is one of the most common benign neoplasms of odontogenic origin. It accounts for 11% of all odontogenic neoplasms/hamartomas (4-6). It is a slow-growing, persistent, and locally aggressive neoplasm of epithelial origin. It affects a wide range of age distribution but is mostly a disease of adults, at an average age of 33, with equal sex distribution. Reports from Africa and India show a male predilection; it also has a predilection for occurrence in black patients (5).
About 85% of ameloblastomas occur in the posterior mandible; most of these occur in the molar-ramus area, and some occur in the anterior mandible. About 15% occur in the maxilla, the vast majority of these in the posterior maxilla, as is the case with our patient. Ameloblastomas of the maxilla tend to occur in patients of an older age, which is also consistent with our patient (4-6), and tend to have a worse prognosis than those of the mandible. They generally recur more and behave more aggressively in terms of early invasion of the maxillary sinus (4, 6). They can also invade the optical nerve and the brain and kill the patient. The notion of ameloblastomas of the maxilla being more aggressive in behavior may be biological in nature such as the histologic type (plexiform ameloblastoma reported to be more aggressive than follicular ameloblastoma) or because of the anatomic location being difficult to reach surgically making complete eradication of a neoplasm a challenge.
Ameloblastoma is rarely described in children. It is characteristically expansile, radiolucent and multilocular in nature. However, it can be unilocular and associated with impacted teeth resembling a dentigerous cyst (4-6). The latter types of ameloblastoma are known to be less aggressive than the multilocular solid lesions. Three clinical types of ameloblastoma are described: the solid type (radiographically multilocular), the cystic type (radiographically unilocular and usually associated with an impacted tooth), and the peripheral type (soft tissue, usually gingival ameloblastoma) (4-6). The present case is of the solid type, although radiographically it is not multilocular in appearance. For more information on the behavior of the various types of ameloblastomas, please read the Clinical Case Discussion in the October 2004 issue of E-PIE.
Ameloblastoma, if not treated, can reach very large sizes, causing facial disfigurement. It loosens, displaces and resorbs adjacent teeth. With the exception of jaw expansion, it is usually asymptomatic unless infected, in which case it can be mildly painful. Parasthesia and anesthesia are extremely rare, unless the lesion is very large in size. Also, ameloblastoma tends to expand rather than perforate the cortical bone; if the latter occurs with extension into the adjacent soft tissue, it has a higher tendency for recurrence and therefore a worse prognosis than cases in which the ameloblastoma is completely encased by bone (4-6).
As mentioned above, three clinical types of ameloblastomas are described, the solid type being the most common. This type is further subtyped histologically into follicular type (which is the most common), acanthomatous type (which is sometimes mistaken for intrabony squamous cell carcinoma), plexiform type (which favors the maxilla over the mandible and is suggested to be more aggressive in behavior), and granular cell, basal cell and desmoplastic types. The last three are the least common (6).
The solid type is treated with complete surgical removal with clean margins through resection or en bloc. Curettage is not recommended for the solid type because it is associated with a higher recurrence rate than the resected counterpart. Resected jaws may require secondary reconstruction (7). Ameloblastoma has an overall good prognosis but is known to have a high recurrence rate, particularly in the posterior maxilla, from inadequate surgery (tumor extending to the surgical margins). Long-term follow-up is required. Recurrence is related to the treatment modality. The histology of this case is not supportive of ameloblastoma.
Sorry! you are incorrect
Central giant cell granuloma is a non-neoplastic process that can occasionally behave in a very aggressive and expansile manner, destroying bone and displacing teeth. Over 60% of CGCG cases occur in patients younger than 30 years of age, with twice as many occurrences in females as in males. CGCG is classified into aggressive and non-aggressive types; the aggressive type tends to occur in younger patients and is known to cause disfiguration, especially after surgery. Over 70% of cases occur in the mandible anterior to the first molar tooth. This lesion has also been described in other cranio-facial and small long bones such as those of the hands and feet (8, 9).
The usual treatment for CGCG is surgery, ranging from curettage and en bloc to resection (9). The latter is used in aggressive and recurring cases (9). Treatment alternatives to surgery have emerged with successful results ranging from steroid injections (10) to calcitonin injections or nasal spray to interferon alfa-2a injections (10), which are administered 2-3 times per week for several months. The latter is the most expensive alternative treatment. The histology of this case is not supportive of CGCG.
Congratulations! You are correct
Odontogenic myxoma should not be mistaken for soft tissue myxoma, which is a relatively common lesion of the soft tissue. It is rare in the mouth; it occurs more commonly in areas such as the heart. When it does occur in the mouth, odontogenic myxoma occurs in the jaw bones, usually in the tooth-bearing areas of the jaw (11-13). It is an uncommon, benign, but locally aggressive neoplasm. Nearly all cases so far have been described in the jaw bones. Therefore, it is of tooth origin, and is believed to be from the mesenchymal portion of a tooth germ, most likely of the dental papilla. It has the potential for extensive bony destruction and extension into the surrounding structures (11-12). It is less common than odontomas and ameloblastomas, constituting around 17% of all odontogenic tumors. For that reason, a pathologist who is not familiar with the histology of a tooth germ can mistake a myxoid dental follicle for an odontogenic myxoma. Almost 75% of odontogenic myxomas occur in patients around 23-30 years of age with a slight female predilection (1:1.5 male-to-female) (11-13). It rarely occurs in patients over 50 or under 10 years of age. It occurs almost equally in the maxilla and mandible with a slight predilection for the posterior mandible. A few cases are described in the ramus and condyle, non-tooth bearing areas.
Odontogenic myxoma is slow-growing, persistent and destructive. Most cases are expansile and can displace and resorb teeth. In the maxilla, they usually invade the maxillary sinuses and at times (though rarely) cross the midline to the opposing sinus. Radiographically, the majority present as expansile and multilocular, though some are unilocular with or without scalloped borders, and rare cases present with a diffuse and mottled appearance which can be mistaken for a malignant neoplasm. Grossly, this lesion is gelatinous in nature, making curettage alone difficult; the more fibrotic odontogenic myxomas (also known as odontogenic myxofibroma or fibromyxoma) have more body and are easier to curette. Histologically, it is made up of loose and delicate fibrous connective tissue. The fibroblasts are stellate and are suspended on a delicate network of collagen fibrils. Immunohistochemistry studies suggest that the spindle-shaped cells constituting this neoplasm have a combined fibroblastic and smooth muscle typing, suggesting that it is of myofibroblastic origin (13). Small blood vessels are present, as are small odontogenic epithelial islands on occasion. Sometimes, this lesion is fibrotic, making it easier to curette.
The treatment of choice is surgical excision ranging from segmental resection with clear bony margins of up 1.5cm to prevent recurrence of the neoplasm. Curettage is with and without cauterization is used for treatment, but is associated with a high recurrence rate. Reconstruction can be immediate or delayed, and can include an autologous bone graft from the anterior or posterior iliac crest. Fibula-free vascular osteocutaneous bone graft is another reconstructive modality, as is distraction osteogenesis. The patient is usually hospitalized for a short period of time following resection and placed on intravenous antibiotics while in the hospital to prevent secondary infection of the surgical site, especially if immediate bone grafting is accomplished. Immediate postoperative follow up is weekly for approximately one month, then monthly for the next five months and twice a year for the next five years.
Treatment
The procedure was performed under general anesthesia. Prior to the incision, arch bars were placed to the left maxillary and left mandibular dentition using 24-gauge circumdental wire. The tumor was removed through an en bloc procedure along with the dentition, teeth numbers 27 through 32. The surgical specimen was radiographed intraoperatively to reveal the bony margins, which were clear for about one centimeter both posteriorly and anteriorly. The surgical site was thoroughly irrigated with bacitracin irrigation and hemostasis was accomplished with small and medium hemoclips as well as bipolar cautery. A prefabricated 2.7 KLS Martin reconstruction plate was pre-adapted to a stereolithic model prior to surgery. This 16-hole plate was adapted to the right ascending ramus of the mandible and the symphyseal-perisymphyseal region and was secured with 12- and 14-millimeter long screws (Fig 3): four on the ascending ramus and four on the perisymphyseal region. The plate was customized and over-bent to allow a transport track for a later reconstruction procedure through a distraction osteogenesis, which took place approximately six weeks after this procedure.
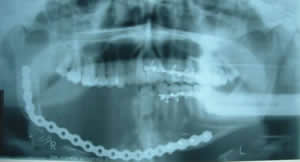
Figure 3. This is a panoramic radiograph representing post-operative changes consistent with resection of the tumor and replacement with a metal plate adapted to the right ascending ramus and the symphyseal-perisymphyseal region.
References
- Shear M. Odontogenic keratocysts: natural history and immunohistochemistry. Oral Maxillofacial Surg Clin N Am. 2003; 15: 347-362.
- Oda D, Rivera V et al. Odontogenic keratocyst: the northwestern USA experience. J Contemp Dent Pract. 2000 Feb 15; 1(2): 60-74.
- Zachariades N, Papanicolaou S et al. Odontogenic keratocysts: Review of the literature and report of sixteen cases. J Oral Maxillofac Surg. 1985; 43: 177-182
- Reichart PA, Philipsen HP. Et al. Ameloblastoma: biological profile of 3677 cases. Eur J Cancer B Oral Oncol 1995;31B:86–99.
- Adekeye EO, McLavery K. Recurrent ameloblastoma of the maxillofacial region. Clinical features and treatment. J Maxillofac Surg 1986;14:153-157.
- Gardner DG. Some current concepts on the pathology of ameloblastomas. J Oral Maxillofac Surg 1996;82:660-669.
- Omandi BI, Guthua SW et al. Maxillary obturator prosthesis rehabilitation following maxillectomy for ameloblastoma: case series of five patients. Int J Prosthodont. 2004;17:464-468.
- Whitaker SB, Vigneswaran N et al. Giant cell lesions of the jaws: evaluation of nucleolar organizer regions of varying behavior. J Oral Pathol Med 1993; 22:402-405.
- Tallan EM, Olsen KD et al. Advanced giant cell granuloma: a twenty-year study. Otolaryngol Head Neck Surg 1994; 110:413-418.
- Carlos R, Sedano HO. Intralesional corticosteroids as an alternative treatment for central giant cell granuloma. Oral Surg Oral Med Oral Pathol 2002; 93:161-166.
- Sharma R, Marwah N. Odontogenic myxoma of the mandible: a case report. Indian J Pathol Microbiol. 2003;46:84-86.
- Simon EN, Merkx MA. Odontogenic myxoma: a clinicopathological study of 33 cases. Int J Oral Maxillofac Surg. 2004;33:333-337.
- Moshiri, S., Oda, D.et al. Odontogenic myxoma: a clinical and immunohistochemical study. J Oral Path 1992;21:401-403.