Gingival swelling between teeth #s 8 & 9
Can you make the correct diagnosis?
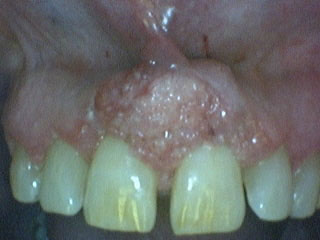
This 61-year-old female was referred by her dentist to a periodontologist for evaluation of an enlarging exophytic, slightly lobular, red and ulcerated gingival swelling between teeth #s 8 & 9 (Figure 1). The area was reported to be slightly painful; it was also reported that the lesion was of cosmetic concern to the patient. She is a mouth-breather and had heavy sub-gingival calculus.
Sorry! you are incorrect
Pyogenic granuloma constitutes 85% of all reactive gingival swellings, representing a profuse mass of vascular granulation tissue (1). It can be induced by local irritants such as excessive plaque, sharp fillings and (as present in this patient) dental calculus; it also sometimes forms in an extraction socket in response to an irritant left in the socket. It can occur anywhere in the oral cavity and skin, especially the tongue, lips, fingers and nail beds (1). In the mouth, it occurs most commonly in the gingiva, especially the maxillary buccal and interproximal gingiva (1-2), as did this patient’s lesion. Occasionally, it may surround the tooth. It is usually highly vascular, fast-growing, exophytic, lobular, sessile, and ulcerated or covered by pseudomembrane. The color changes from red to pink when it starts to heal. It occurs at any age and sex with a slight predilection for young females; it affects 1% of pregnant females. Pyogenic granuloma is usually painless except during eating, when bleeding and pain is described (1). Histologically, it presents as a mass of loose and vascular granulation tissue, usually with ulcerated or eroded surface epithelium and many inflammatory cells. A range of treatment modalities are available, including excision with removal of the local irritant, laser surgery, or intralesional injection with absolute alcohol, steroids or botulinum toxin (2-3). Scaling and polishing prior to surgical removal helps shrink the lesion. The prognosis is good, although recurrence is possible, especially during pregnancy. While the clinical presentation of this case is consistent with pyogenic granuloma, the histology is not.
Sorry! you are incorrect
The following are some of the medications known to have the potential to produce gingival hyperplasia: calcium channel blockers (amlodipine, bepridil, diltiazem, felodipine, isradipine, nicardipine, nifedipine, nimodipine, nisoldipine, nitrendipine, oxidipine, verapamil), other dihydropyridines (bleomycin), cyclosporine, phenytoin, and sodium valproate.
Gingival hyperplasia (GH) caused by drug intake has been recognized since the 1960s and has been associated with phenytoin/dilantin, taken for seizures, affecting up to 50% of patients. Since the early 1980s gingival hyperplasia associated with several calcium channel blockers, especially nifedipine (affecting up to 20% of patients), has been described (4-5). Since the beginning of the era of organ transplants, GH has also been described in association with cyclosporine (affecting up to 27% of patients), a potent immunosuppressant used in organ transplant patients. It is important to note that millions of patients are on one or more of the three aforementioned drugs, but not all develop GH (5-6); therefore, those patients at a particular risk of developing GH must be recognized. There is no age or sex predilection, although Dilantin-induced GH is more common in children and calcium channel blocker-induced GH is more common in adults as a result of the fact that epilepsy and hypertension are more common in those respective age groups. Gingival hyperplasia occurs more commonly on the facial or labial gingiva and in the interdental papilla area involving multiple teeth (4-6). Dilantin- and nifedipine-induced GH tend to be red and cyclosporine-induced GH tends to be pale pink to white. Treatment is a multifactorial system in which the patient’s physician should be consulted for a possibility of change of medication or reduction in dose concentration. Sometimes it is contraindicated, as is the case with cyclosporine. Oral hygiene instructions and more frequent cleaning visits are also suggested. The clinical background in this case may be suggestive of drug-induced gingival hyperplasia, but the histology is not.
Sorry! you are incorrect
Mouth breathing is breathing with the mouth open. Two of the main reasons for mouth breathing are chronic nasal obstruction and a high, narrow palate (7). It leads to dryness of the mucosa, especially that of the anterior labial maxillary gingiva, and sometimes that of the mandibular anterior labial gingiva. It can occur at any age (7). The early manifestation of mouth breathing-induced gingivitis is redness of the labial marginal and interdental gingival; with time, diffuse gingival redness, swelling and bleeding take place. The best treatment is to treat the cause of the mouth breathing, such as treating an obstructive nasal condition if present. For temporary results, the gingiva can be protected with dressings such as Vaseline. The histology of mouth breathing-induced gingivitis is similar to any chronic gingivitis in which inflammatory cells, fibrosis and congested blood vessels would be identified. Neither the history nor the clinical presentation of this lesion is supportive of mouth breathing-induced gingivitis.
Congratulations! You are correct
Squamous cell carcinoma of the gingiva is uncommon, especially in a non-smoker and social alcohol user. Oral squamous cell carcinoma (OSCC) of the mouth is a highly aggressive neoplasm that currently ranks as the fifth most common malignant neoplasm worldwide and accounts for an estimated 90% of oral malignancies (8). Oral SCC occurs predominantly in males over the age of 40 years, with an observed male-to-female ratio of 2:1 generally and 1.4:1 in the USA (8-9). Excluding the outer lip, the most common sites (in decreasing order) are ventral and lateral surfaces of tongue (25-50%), floor of mouth (15%), gingiva (12%) and palate (9%). The buccal mucosa and retromolar pad areas (3%) have a relatively low incidence of occurrence (10) unless the patient is a chronic smokeless tobacco user. Oral SCC varies in presentation from deceptively innocent-looking to obviously malignant. It may present as a non-healing ulcer, or as red, white or mixed red-and-white lesions. Characteristic signs of oral SCC are non-healing ulcer, ulcer with rolled borders, fungation, fixation and induration. Rarely, OSCC may present as unexplained asymptomatic lateral neck lymphadenopathy (8-10). Oral SCC is most commonly associated with chemically induced mutagenesis, specifically tobacco and alcohol use (11).
Tobacco use is described in over 75% of oral SCC patients (8, 11). Tobacco and alcohol have been shown to act synergistically in the development of OSCC (12). Human papilloma virus (HPV) has also been found to have a high prevalence in oral cancer, especially in younger patients with no history of tobacco use (12). Other factors include poor oral hygiene, syphilis, chronic candidiasis, iron and dietary deficiencies, herpes simplex and various other immunologic factors (12), and lichen planus—especially the persistent erosive lichen planus (13).
Determination of the prognosis of OSCC is based on its clinical stage and histological classification. Although OSCC is a diagnosis made by histology, surgeons tend to depend exclusively on the TNM classification system for clinical staging and treatment decisions (14). Prognosis is dependent on the TNM staging system; the most important prognostic sign is the presence or absence of metastases at the time of diagnosis. The prognosis thus improves when the lesion is detected early. Oral SCC patients die mainly of infection due to lowered resistance or of hemorrhage if the tumor erodes through one of the main blood vessels.
References
- Fantasia JE, Damm DD. Red nodular lesion of tongue. Pyogenic granuloma. Gen Dent. 2003 Mar-Apr;51(2):190, 194.
- Ichimiya M, Yoshikawa Y, Hamamoto Y, Muto M. Successful treatment of pyogenic granuloma with injection of absolute ethanol. J Dermatol. 2004 Apr;31(4):342-4.
- Pham J, Yin S, Morgan M, Stucker F, Nathan CA. Botulinum toxin: helpful adjunct to early resolution of laryngeal granulomas. J Laryngol Otol. 2004 Oct;118(10):781-5.
- Jaiarj N. J Mass Dent Soc. 2003 Fall;52(3):16-20.
- Varnfield M SADJ. 2000 Nov;55(11):632-41.
- Abdollahi M J Contemp Dent Pract. 2003 Feb 15;4(1):10-31.
- Wagaiyu EG, Ashley FP. Mouthbreathing, lip seal and upper lip coverage and their relationship with gingival inflammation in 11-14 year-old schoolchildren. J Clin Periodontol. 1991 Oct;18(9):698-702.
- Food, Nutrition, and the Prevention of Cancer: a global perspective. World Health Organization. 1997.
- Examinations for oral cancer – United States. MMWR. Morb.MortalWkly Rep. 43:198 (1992).
- Bundgaard T, S Bentzen, et al. Histopathologic, stereologic, Epidemiologic, and clinical parameters in the prognostic evaluation of squamous cell carcinoma of the oral cavity. Head & Neck. 18:142-152 (1996).
- Barasch A, DE Morse, et al. Smoking, gender, and age as risk factors for site-specific intraoral squamous cell carcinoma. Cancer 73:509-513 (1994).
- Syrjanen SM, KJ Syrjanen et al. Human papillomavirus (HPV) DNA sequences in oral precancerous lesions and squamous cell carcinoma demonstrated by in situ hybridization. J Oral Pathol. 17:273 (1988).
- Holmstrup P, JJ Thorn, et al. Malignant development of lichen planus-affected oral mucosa. J Oral Pathol. 17:219-25 (1988).
- American Joint Committee for Cancer Staging and End Result Reporting. (1983).